Breakthrough in selective CO2 capturing materials; could make bioenergy carbon-negative
Chemists from the University of California - Los Angeles (UCLA) have made a major advancement in the development of CO2 capturing materials, which they report in the Feb. 15 issue of the journal Science. The scientists have demonstrated that they can successfully isolate and capture carbon dioxide with a class of new materials known as zeolitic imidazolate frameworks (ZIFs). Their findings could lead to power plants efficiently capturing the greenhouse gas without using toxic materials, after which it can be stored in geological formations. The new materials make carbon capture less energy demanding, and can store up to five times as much CO2 than porous carbon materials being designed for the same task.
When carbon capture and storage (CCS) is coupled to biomass energy production, negative emissions are the result. Such carbon-negative bioenergy systems - which actively remove CO2 from the atmosphere - are the most radical tool in the climate fight. They come in the form of decarbonised bio-electricity or biohydrogen. Ordinary renewables like solar, wind, non-CCS bioenergy or hydropower are all 'carbon-neutral' at best, that is, they do not add new or only modest amounts of CO2 to the atmosphere, but they do not take the greenhouse gas out if either. Bioenergy with carbon storage does. The difference can be very significant: whereas the lifecycle carbon emissions of ordinary renewables and nuclear range between +10 to +100 grams of CO2eq per kWh, bioenergy coupled to CCS can yield as high as -1000gCO2/kWh (that is: minus 1000 grams, hence "negative emissions"), and thus clean up the atmosphere by removing CO2 from the past.
Efficient and affordable CO2 capturing technologies are needed to make such carbon-negative bioenergy systems feasible (see list of references below for an overview of developments). The UCLA breakthrough, made at professor Omar Yaghi's lab, goes a long way in meeting this need:
Rahul Banerjee, a UCLA postdoctoral research scholar in chemistry and Anh Phan, a UCLA graduate student in chemistry, both of whom work in Yaghi's laboratory, synthesized 25 ZIF crystal structures and demonstrated that three of them have high selectivity for capturing carbon dioxide (ZIF-68, ZIF-69, ZIF-70).
The selectivity of ZIFs to carbon dioxide is unparalleled by any other material, said Yaghi, who directs of UCLA's Center for Reticular Chemistry and is a member of the California NanoSystems Institute at UCLA. Rahul and Anh were so successful at making new ZIFs that, for the purposes of reporting the results, Yaghi had to ask them to stop.
The inside of a ZIF can store gas molecules. Flaps that behave like the chemical equivalent of a revolving door allow certain molecules — in this case, carbon dioxide — to pass through and enter the reservoir while blocking larger molecules or molecules of different shapes:
 energy :: sustainability :: biomass :: bioenergy :: biofuels :: carbon capture and storage :: decarbonisation :: geosequestration :: carbon-negative :: negative emissions :: bio-energy with carbon storage :: zeolites :: climate change ::
energy :: sustainability :: biomass :: bioenergy :: biofuels :: carbon capture and storage :: decarbonisation :: geosequestration :: carbon-negative :: negative emissions :: bio-energy with carbon storage :: zeolites :: climate change ::
Currently, the process of capturing carbon dioxide emissions from power plants involves the use of toxic materials and requires 20 to 30 percent of the plant's energy output, Yaghi said. By contrast, ZIFs can pluck carbon dioxide from other gases that are emitted and can store five times more carbon dioxide than the porous carbon materials that represent the current state-of-art. For each liter of ZIF, you can hold 83 liters of carbon dioxide.
On a fundamental level, the invention of ZIFs has addressed two major challenges in zeolite science. Zeolites are stable, porous minerals made of aluminum, silicon and oxygen that are employed in petroleum refining and are used in detergents and other products. Yaghi's group has succeeded in replacing what would have been aluminum or silicon with metal ions like zinc and cobalt, and the bridging oxygen with imidazolate to yield ZIF materials, whose structures can now be designed in functionality and metrics.
Banerjee and Anh automated the process of synthesis. Instead of mixing the chemicals one reaction at a time and achieving perhaps several reactions per day, they were able to perform 200 reactions in less than an hour. The pair ran 9,600 microreactions and from those reactions uncovered 25 new structures. The scientists say they keep producing new crystals of ZIFs every day.
Co-authors are Bo Wang, a UCLA graduate student in chemistry in Yaghi's laboratory; Carolyn Knobler and Hiroyasu Furukawa of the Center for Reticular Chemistry at the UCLA's California NanoSystems Institute; and Michael O'Keeffe of Arizona State University's department of chemistry and biochemistry.
BASF, a global chemical company based in Germany, funded the synthesis of the materials, and the U.S. Department of Energy funded the absorption and separation studies of carbon dioxide.
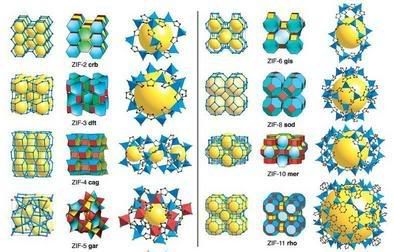
Image: The single crystal x-ray structures of ZIFs. (Left and Center) In each row, the net is shown as a stick diagram (Left) and as a tiling (Center). (Right) The largest cage in each ZIF is shown with ZnN4 tetrahedra in blue, and, for ZIF-5, InN6 octahedra in red. H atoms are omitted for clarity. Credit: Yaghi Lab.
References:
Eurekalert: New materials can selectively capture carbon dioxide, UCLA chemists report - February 15, 2007.
The reference to the Science article was not yet available at the time of writing; we will update this article as soon as it does.
Yaghi's team did publish about ZIFs earlier:
Hideki Hayashi, Adrien P. Côté, Hiroyasu Furukawa, Michael O'Keeffe & Omar M. Yaghi, "Zeolite A imidazolate frameworks" [*.pdf - at Yaghi Lab], Nature Materials 6, 501 - 506 (2007), Published online: 27 May 2007 | doi:10.1038/nmat1927
Omar M. Yaghi, et. al. "Exceptional chemical and thermal stability of zeolitic imidazolate frameworks" [*.pdf - at Yaghi Lab], PNAS, July 5, 2006, vol. 103, no. 27, 10186-10191, DOI: 10.1073/pnas.0602439103
Yaghi Laboratory.
Scientific literature on negative emissions from biomass:
H. Audus and P. Freund, "Climate Change Mitigation by Biomass Gasificiation Combined with CO2 Capture and Storage", IEA Greenhouse Gas R&D Programme.
James S. Rhodesa and David W. Keithb, "Engineering economic analysis of biomass IGCC with carbon capture and storage", Biomass and Bioenergy, Volume 29, Issue 6, December 2005, Pages 440-450.
Noim Uddin and Leonardo Barreto, "Biomass-fired cogeneration systems with CO2 capture and storage", Renewable Energy, Volume 32, Issue 6, May 2007, Pages 1006-1019, doi:10.1016/j.renene.2006.04.009
Christian Azar, Kristian Lindgren, Eric Larson and Kenneth Möllersten, "Carbon Capture and Storage From Fossil Fuels and Biomass – Costs and Potential Role in Stabilizing the Atmosphere", Climatic Change, Volume 74, Numbers 1-3 / January, 2006, DOI 10.1007/s10584-005-3484-7
Further reading on negative emissions bioenergy and biofuels, and carbon capture techniques:
Peter Read and Jonathan Lermit, "Bio-Energy with Carbon Storage (BECS): a Sequential Decision Approach to the threat of Abrupt Climate Change", Energy, Volume 30, Issue 14, November 2005, Pages 2654-2671.
Stefan Grönkvist, Kenneth Möllersten, Kim Pingoud, "Equal Opportunity for Biomass in Greenhouse Gas Accounting of CO2 Capture and Storage: A Step Towards More Cost-Effective Climate Change Mitigation Regimes", Mitigation and Adaptation Strategies for Global Change, Volume 11, Numbers 5-6 / September, 2006, DOI 10.1007/s11027-006-9034-9
Biopact: Commission supports carbon capture & storage - negative emissions from bioenergy on the horizon - January 23, 2008
Biopact: The strange world of carbon-negative bioenergy: the more you drive your car, the more you tackle climate change - October 29, 2007
Biopact: "A closer look at the revolutionary coal+biomass-to-liquids with carbon storage project" - September 13, 2007
Biopact: New plastic-based, nano-engineered CO2 capturing membrane developed - September 19, 2007
Biopact: Plastic membrane to bring down cost of carbon capture - August 15, 2007
Biopact: Pre-combustion CO2 capture from biogas - the way forward? - March 31, 2007
Biopact: Towards carbon-negative biofuels: US DOE awards $66.7 million for large-scale CO2 capture and storage from ethanol plant - December 19, 2007
Biopact: Biochar and carbon-negative bioenergy: boosts crop yields, fights climate change and reduces deforestation - January 28, 2008
When carbon capture and storage (CCS) is coupled to biomass energy production, negative emissions are the result. Such carbon-negative bioenergy systems - which actively remove CO2 from the atmosphere - are the most radical tool in the climate fight. They come in the form of decarbonised bio-electricity or biohydrogen. Ordinary renewables like solar, wind, non-CCS bioenergy or hydropower are all 'carbon-neutral' at best, that is, they do not add new or only modest amounts of CO2 to the atmosphere, but they do not take the greenhouse gas out if either. Bioenergy with carbon storage does. The difference can be very significant: whereas the lifecycle carbon emissions of ordinary renewables and nuclear range between +10 to +100 grams of CO2eq per kWh, bioenergy coupled to CCS can yield as high as -1000gCO2/kWh (that is: minus 1000 grams, hence "negative emissions"), and thus clean up the atmosphere by removing CO2 from the past.
Efficient and affordable CO2 capturing technologies are needed to make such carbon-negative bioenergy systems feasible (see list of references below for an overview of developments). The UCLA breakthrough, made at professor Omar Yaghi's lab, goes a long way in meeting this need:
The technical challenge of selectively removing carbon dioxide has been overcome. Now we have structures that can be tailored precisely to capture carbon dioxide and store it like a reservoir, as we have demonstrated. No carbon dioxide escapes. Nothing escapes — unless you want it to do so. We believe this to be a turning point in capturing carbon dioxide before it reaches the atmosphere. - Omar M. Yaghi, UCLA's Christopher S. Foote Professor of Chemistry and co-author of the Science paperThe carbon dioxide is captured using a new class of materials designed by Yaghi and his group called zeolitic imidazolate frameworks, or ZIFs. These are porous and chemically robust structures, with large surface areas, that can be heated to high temperatures without decomposition and boiled in water or organic solvents for a week and still remain stable.
Rahul Banerjee, a UCLA postdoctoral research scholar in chemistry and Anh Phan, a UCLA graduate student in chemistry, both of whom work in Yaghi's laboratory, synthesized 25 ZIF crystal structures and demonstrated that three of them have high selectivity for capturing carbon dioxide (ZIF-68, ZIF-69, ZIF-70).
The selectivity of ZIFs to carbon dioxide is unparalleled by any other material, said Yaghi, who directs of UCLA's Center for Reticular Chemistry and is a member of the California NanoSystems Institute at UCLA. Rahul and Anh were so successful at making new ZIFs that, for the purposes of reporting the results, Yaghi had to ask them to stop.
The inside of a ZIF can store gas molecules. Flaps that behave like the chemical equivalent of a revolving door allow certain molecules — in this case, carbon dioxide — to pass through and enter the reservoir while blocking larger molecules or molecules of different shapes:
We can screen and select the one type of molecule we want to captureThe beauty of the chemistry is that we have the freedom to choose what kind of door we want and to control what goes through the door. - Anh Phan, a UCLA graduate student in chemistry, developer of selectiv ZIFsIn ZIFs 68, 69 and 70, Banerjee and Phan emptied the pores, creating an open framework. They then subjected the material to streams of gases - carbon dioxide and carbon monoxide, for example, and another stream of carbon dioxide and nitrogen — and were able to capture only the carbon dioxide. They are testing other ZIFs for various applications:
 energy :: sustainability :: biomass :: bioenergy :: biofuels :: carbon capture and storage :: decarbonisation :: geosequestration :: carbon-negative :: negative emissions :: bio-energy with carbon storage :: zeolites :: climate change ::
energy :: sustainability :: biomass :: bioenergy :: biofuels :: carbon capture and storage :: decarbonisation :: geosequestration :: carbon-negative :: negative emissions :: bio-energy with carbon storage :: zeolites :: climate change :: Currently, the process of capturing carbon dioxide emissions from power plants involves the use of toxic materials and requires 20 to 30 percent of the plant's energy output, Yaghi said. By contrast, ZIFs can pluck carbon dioxide from other gases that are emitted and can store five times more carbon dioxide than the porous carbon materials that represent the current state-of-art. For each liter of ZIF, you can hold 83 liters of carbon dioxide.
On a fundamental level, the invention of ZIFs has addressed two major challenges in zeolite science. Zeolites are stable, porous minerals made of aluminum, silicon and oxygen that are employed in petroleum refining and are used in detergents and other products. Yaghi's group has succeeded in replacing what would have been aluminum or silicon with metal ions like zinc and cobalt, and the bridging oxygen with imidazolate to yield ZIF materials, whose structures can now be designed in functionality and metrics.
Banerjee and Anh automated the process of synthesis. Instead of mixing the chemicals one reaction at a time and achieving perhaps several reactions per day, they were able to perform 200 reactions in less than an hour. The pair ran 9,600 microreactions and from those reactions uncovered 25 new structures. The scientists say they keep producing new crystals of ZIFs every day.
Co-authors are Bo Wang, a UCLA graduate student in chemistry in Yaghi's laboratory; Carolyn Knobler and Hiroyasu Furukawa of the Center for Reticular Chemistry at the UCLA's California NanoSystems Institute; and Michael O'Keeffe of Arizona State University's department of chemistry and biochemistry.
BASF, a global chemical company based in Germany, funded the synthesis of the materials, and the U.S. Department of Energy funded the absorption and separation studies of carbon dioxide.
Image: The single crystal x-ray structures of ZIFs. (Left and Center) In each row, the net is shown as a stick diagram (Left) and as a tiling (Center). (Right) The largest cage in each ZIF is shown with ZnN4 tetrahedra in blue, and, for ZIF-5, InN6 octahedra in red. H atoms are omitted for clarity. Credit: Yaghi Lab.
References:
Eurekalert: New materials can selectively capture carbon dioxide, UCLA chemists report - February 15, 2007.
The reference to the Science article was not yet available at the time of writing; we will update this article as soon as it does.
Yaghi's team did publish about ZIFs earlier:
Hideki Hayashi, Adrien P. Côté, Hiroyasu Furukawa, Michael O'Keeffe & Omar M. Yaghi, "Zeolite A imidazolate frameworks" [*.pdf - at Yaghi Lab], Nature Materials 6, 501 - 506 (2007), Published online: 27 May 2007 | doi:10.1038/nmat1927
Omar M. Yaghi, et. al. "Exceptional chemical and thermal stability of zeolitic imidazolate frameworks" [*.pdf - at Yaghi Lab], PNAS, July 5, 2006, vol. 103, no. 27, 10186-10191, DOI: 10.1073/pnas.0602439103
Yaghi Laboratory.
Scientific literature on negative emissions from biomass:
H. Audus and P. Freund, "Climate Change Mitigation by Biomass Gasificiation Combined with CO2 Capture and Storage", IEA Greenhouse Gas R&D Programme.
James S. Rhodesa and David W. Keithb, "Engineering economic analysis of biomass IGCC with carbon capture and storage", Biomass and Bioenergy, Volume 29, Issue 6, December 2005, Pages 440-450.
Noim Uddin and Leonardo Barreto, "Biomass-fired cogeneration systems with CO2 capture and storage", Renewable Energy, Volume 32, Issue 6, May 2007, Pages 1006-1019, doi:10.1016/j.renene.2006.04.009
Christian Azar, Kristian Lindgren, Eric Larson and Kenneth Möllersten, "Carbon Capture and Storage From Fossil Fuels and Biomass – Costs and Potential Role in Stabilizing the Atmosphere", Climatic Change, Volume 74, Numbers 1-3 / January, 2006, DOI 10.1007/s10584-005-3484-7
Further reading on negative emissions bioenergy and biofuels, and carbon capture techniques:
Peter Read and Jonathan Lermit, "Bio-Energy with Carbon Storage (BECS): a Sequential Decision Approach to the threat of Abrupt Climate Change", Energy, Volume 30, Issue 14, November 2005, Pages 2654-2671.
Stefan Grönkvist, Kenneth Möllersten, Kim Pingoud, "Equal Opportunity for Biomass in Greenhouse Gas Accounting of CO2 Capture and Storage: A Step Towards More Cost-Effective Climate Change Mitigation Regimes", Mitigation and Adaptation Strategies for Global Change, Volume 11, Numbers 5-6 / September, 2006, DOI 10.1007/s11027-006-9034-9
Biopact: Commission supports carbon capture & storage - negative emissions from bioenergy on the horizon - January 23, 2008
Biopact: The strange world of carbon-negative bioenergy: the more you drive your car, the more you tackle climate change - October 29, 2007
Biopact: "A closer look at the revolutionary coal+biomass-to-liquids with carbon storage project" - September 13, 2007
Biopact: New plastic-based, nano-engineered CO2 capturing membrane developed - September 19, 2007
Biopact: Plastic membrane to bring down cost of carbon capture - August 15, 2007
Biopact: Pre-combustion CO2 capture from biogas - the way forward? - March 31, 2007
Biopact: Towards carbon-negative biofuels: US DOE awards $66.7 million for large-scale CO2 capture and storage from ethanol plant - December 19, 2007
Biopact: Biochar and carbon-negative bioenergy: boosts crop yields, fights climate change and reduces deforestation - January 28, 2008
 --------------
--------------

 The Andersons, Inc. and Marathon Oil Corporation today jointly announced ethanol production has begun at their 110-million gallon ethanol plant located in Greenville, Ohio. Along with the 110 million gallons of ethanol, the plant annually will produce 350,000 tons of distillers dried grains, an animal feed ingredient.
The Andersons, Inc. and Marathon Oil Corporation today jointly announced ethanol production has begun at their 110-million gallon ethanol plant located in Greenville, Ohio. Along with the 110 million gallons of ethanol, the plant annually will produce 350,000 tons of distillers dried grains, an animal feed ingredient.








2 Comments:
Wonderful! Hope this successfully reduces to practice...
CO2 is a valuable reactant in a lot of chemical processes. If they can capture and concentrate the CO2 in pure form, it will be of some value.
The biosphere itself does not want significantly lowered CO2 pressures, however. If you halved CO2 levels, the biosphere would be strained to produce at current rates. In fact, doubling atmospheric CO2 would be better for the biosphere than halving atmospheric CO2 would be.
That goes against conventional wisdom, but conventional wisdom is generally a load of crap.
Post a Comment
Links to this post:
Create a Link
<< Home